Open Trials
- Details
|
|
A multicentre single-arm phase II trial of amivantamab, lazertinib plus bevacizumab in patients with EGFR-mutant advanced NSCLC with progression on previous third-generation EGFR-TKI.
AMAZE-lung: Amivantamab, lazertinib and bevacizumab in patients with EGFR-mutant advanced non-small cell lung cancer with progression on previous third-generation EGFR-TKI
The AMAZE-lung study aims to test the efficacy of amivantamab, bevacizumab and lazertinib in patients with EGFR-mutant advanced NSCLC.
Trial Scheme
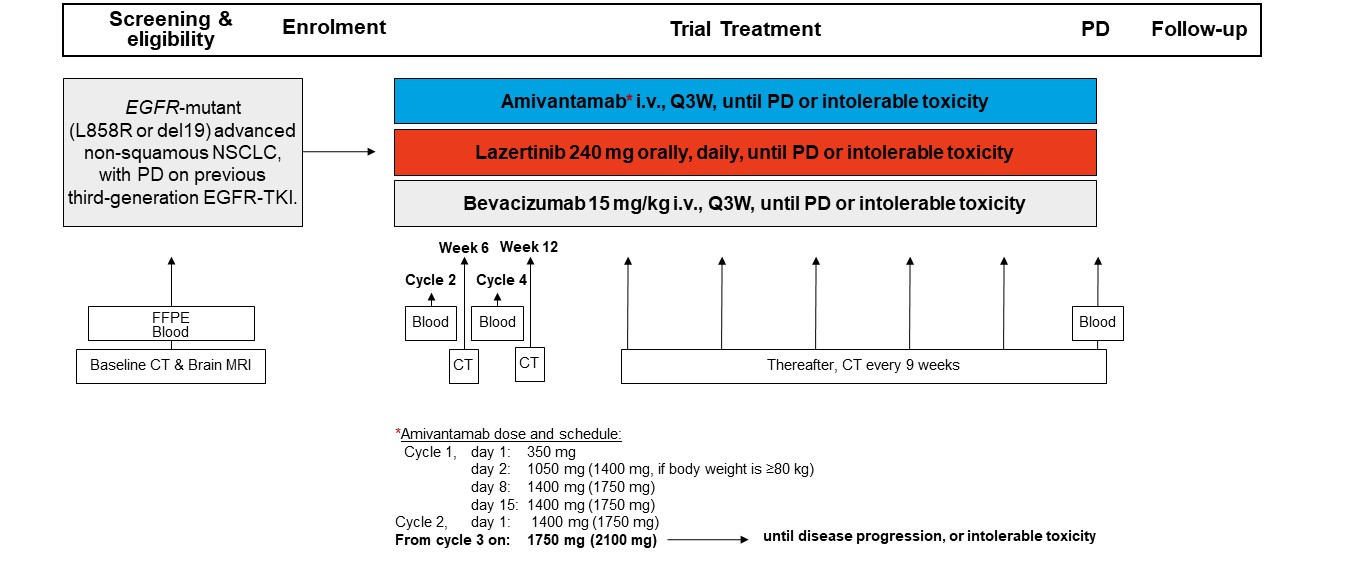
| Primary Endpoint: | Objective response rate (ORR), investigator assessed, at 12 weeks according to RECIST v1.1 |
| Secondary Endpoints: |
Duration of response (DoR) Progression-free survival (PFS) according to RECIST v1.1 Disease control rate (DCR) according to RECIST v1.1 Overall survival (OS) Safety and tolerability (CTCAE v5.0) |
| Target Sample Size: | 60 enrolled patients |
| Protocol Release Date: | 14 September 2022 |
Trial Organisation |
|
| Trial Chair: | Ross Soo, Singapore |
| Trial Co-Chair: |
Sanjay Popat, London, United Kingdom |
| Sponsor: | ETOP IBCSG Partners Foundation |
| Coordinating Group: | ETOP IBCSG Partners Foundation |
| Participating Groups: | Spanish Lung Cancer Group (SLCG) |
| Participating Countries: |
France, Italy, the Netherlands, Singapore, South Korea, Spain, Switzerland, United Kingdom |
| Registrations: |
EudraCT number: 2021-002337-42 clinicaltrials.gov: NCT05601973 |
Contact
Uli Kodjadjiku (Clinical Trial Manager)
ETOP IBCSG Partners Foundation
Effingerstrasse 33
3008 Bern, Switzerland
- Details
A single-arm phase II trial of the addition of niraparib to anti-PD-L1 antibody maintenance in patients with SLFN11-positive, extensive-disease small cell lung cancer.
Niraparib added to anti-PD-L1 antibody maintenance in SLFN11-positive, extensive-disease SCLC
An international, multicentre, single-arm phase II trial in patients with extensive-disease small cell lung cancer and high SLFN11-expression who have not progressed during first-line standard chemo-immunotherapy and are planned for maintenance therapy with immune-checkpoint inhibition .
Trial Scheme
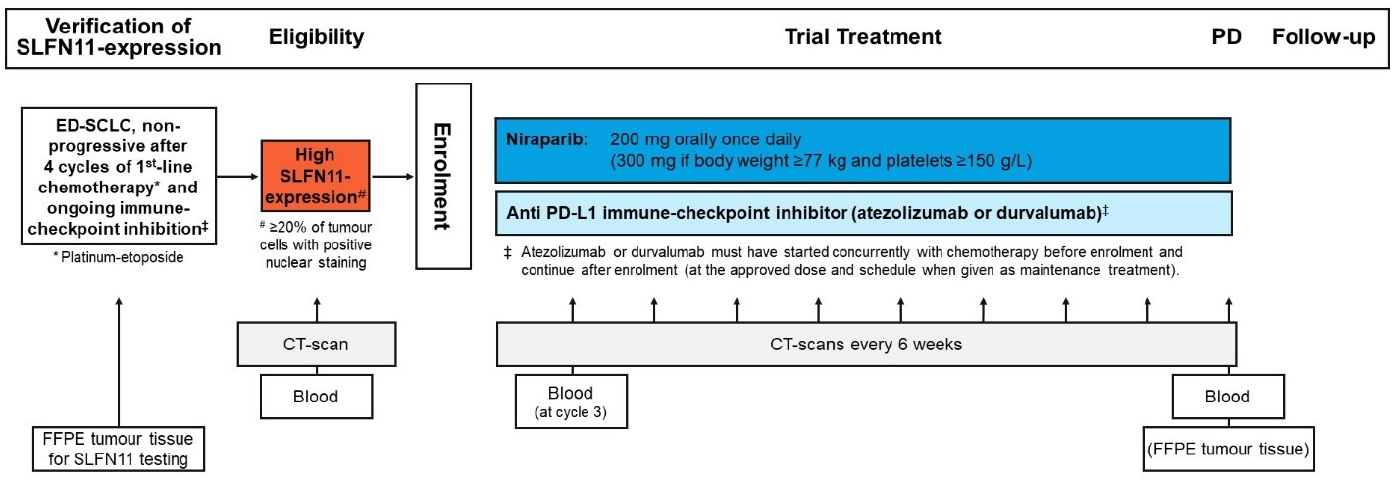
| Primary Endpoint: | Progression-free survival (PFS) rate at 3 months by investigator assessment (according to RECIST v1.1) |
| Secondary Endpoints: |
Progression-free survival (PFS) Overall survival (OS) Disease control rate (DCR) by investigator assessment (according to RECIST v1.1) Adverse events according to CTCAE v5.0 |
| Target Sample Size: | 44 enrolled patients |
| Protocol Release Date: | 21 February 2023 |
Trial Organisation |
|
| Trial Chair: |
Markus Joerger, St. Gallen, Switzerland |
| Trial Co-Chair: |
Antonio Passaro, Milano, Italy |
| Sponsor: |
ETOP IBCSG Partners Foundation |
| Coordinating Group: |
ETOP IBCSG Partners Foundation |
| Participating Groups: | SAKK and SLCG |
| Participating Countries: |
France, Italy, Romania, Switzerland and Spain |
| Registrations: |
EU CT number: 2022-502092-33 clinicaltrials.gov: NCT05718323 |
Contact
Virginia Rodriguez Martinez (Clinical Trial Manager)
ETOP IBCSG Partners Foundation
Effingerstrasse 33
3008 Bern, Switzerland
- Details
|
|
A multicentre randomised open-label phase III study of stereotactic radiosurgery, in addition to standard systemic therapy for patients with metastatic melanoma or newly diagnosed metastatic NSCLC and asymptomatic or oligo-symptomatic brain metastases.
USZ-STRIKE: Immunotherapy or targeted therapy with or without stereotactic radiosurgery for patients with brain metastases from melanoma or non-small cell lung cancer
The trial aims to assess the efficacy of standard systemic treatment plus stereotactic radiosurgery in patients with newly diagnosed brain metastases from melanoma or NSCLC.
Trial Scheme
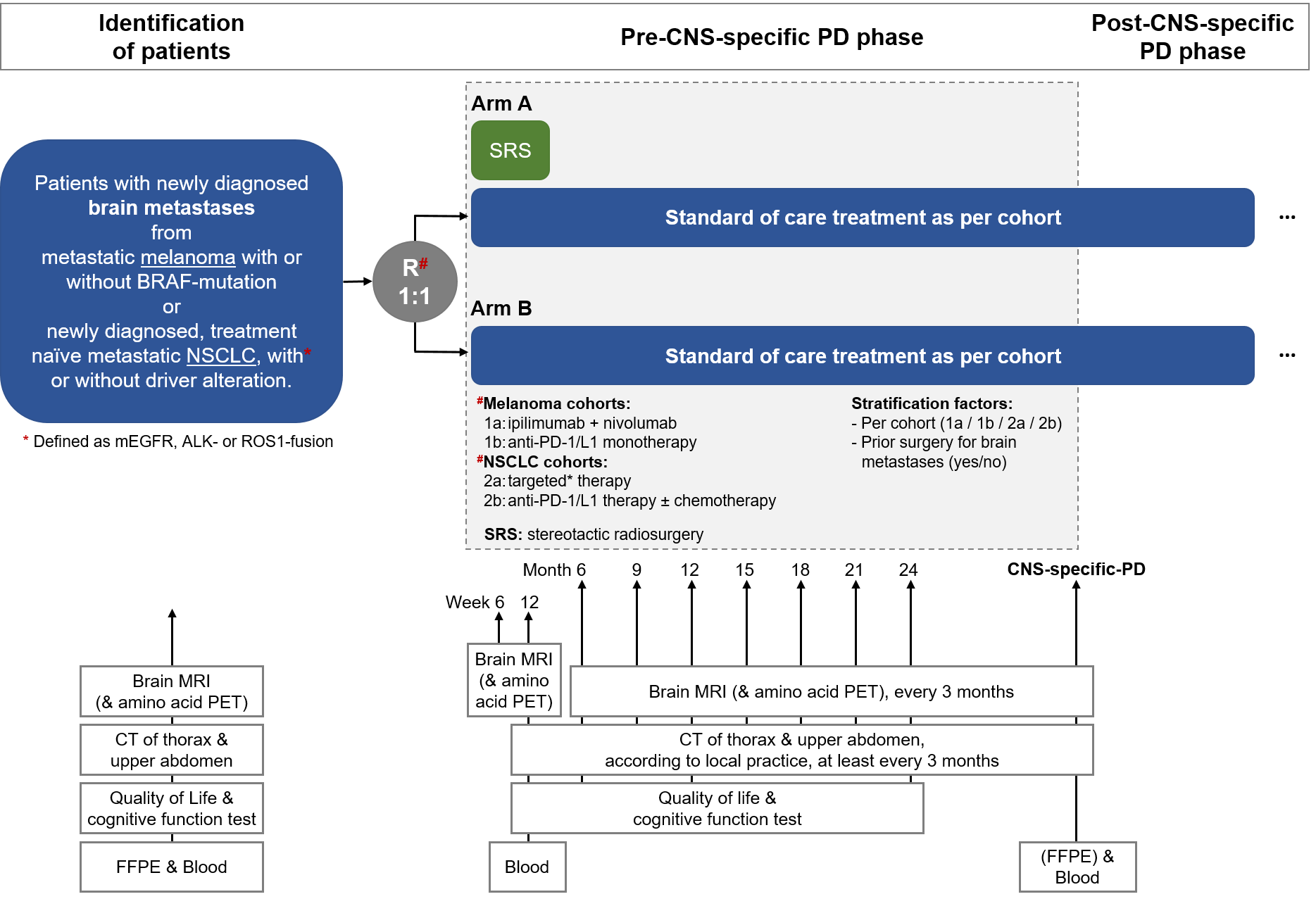
| Primary Endpoint: | CNS-specific PFS, locally assessed as per iRANO criteria |
| Secondary Endpoints: |
CNS-specific PFS per tumour cohort, locally assessed as per iRANO criteria CNS-specific PFS, overall and per tumour cohort, centrally assessed as per iRANO criteria Objective CNS-response rate, centrally assessed as per iRANO criteria Duration of CNS-response Pattern of CNS-specific progression (local versus distant progression) Extra-CNS progression, locally assessed as per RECIST v1.1 Incidence of radio-necrosis and pseudo-progression in the CNS OS, overall and per tumour cohort Neurocognitive function Quality of life and functional independence Toxicity by CTCAE v5 |
| Target Sample Size: | 190 randomised patients |
| Protocol Release Date: | 14 February 2022 |
Trial Organisation |
|
| Trial Chair: | Michael Weller |
| Trial Co-Chairs: | Rolf Stahel |
| Sponsor: | ETOP IBCSG Partners Foundation |
| Coordinating Group: | ETOP IBCSG Partners Foundation |
| Participating Countries: |
Italy, the Netherlands, Switzerland, and United Kingdom |
| Registrations: | NCT05522660 |
Contact
Julien Orgül (Clinical Trial Manager)
ETOP IBCSG Partners Foundation
Effingerstrasse 33
3008 Bern, Switzerland
- Details
|
|
A multicentre, single-arm phase II trial of adagrasib in patients with KRASG12C-mutant NSCLC, including the elderly (≥70 years) or patients with poor performance status.
ADEPPT: Adagrasib in patients with KRASG12C-mutant NSCLC who are elderly or have poor performance status
The aim of the trial is to assess the clinical efficacy of adagrasib treatment, in terms of objective response, in patients with KRASG12C‑mutant NSCLC, including the elderly (≥70 years) or patients with poor performance status (ECOG PS=2).
Trial Scheme
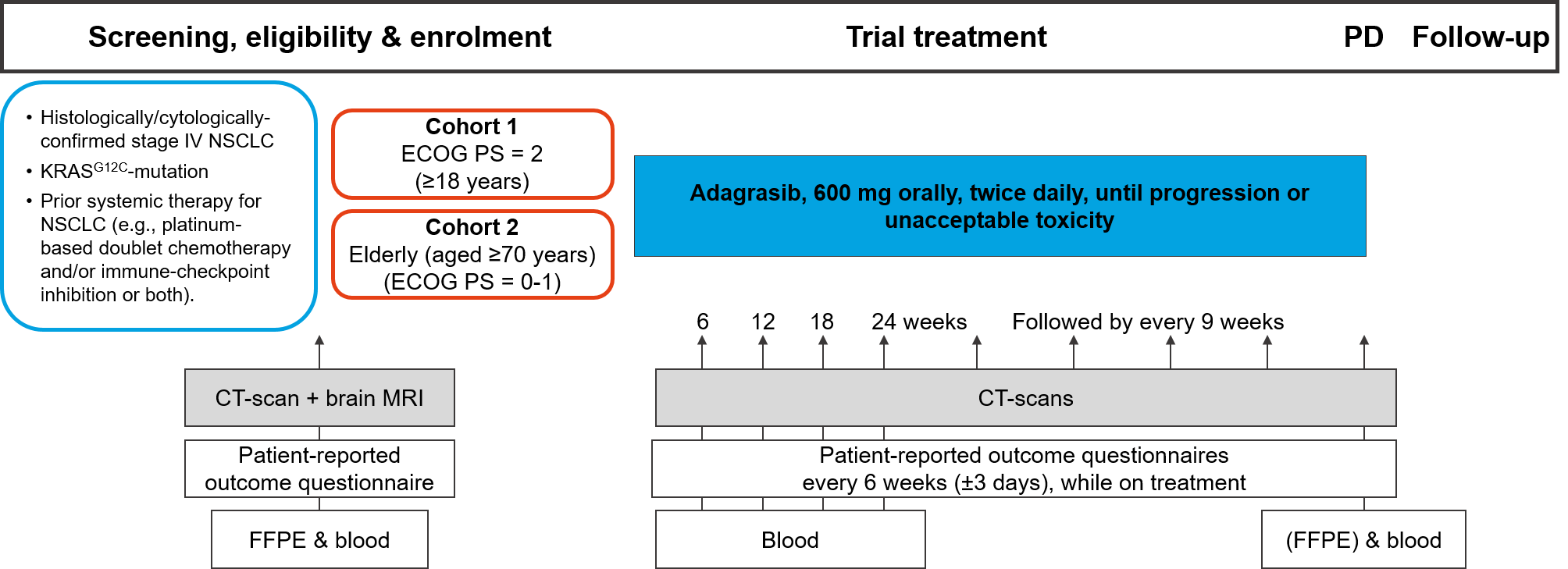
| Primary Endpoint: | Objective Response Rate (ORR) per RECIST v1.1, assessed at 12 weeks |
| Secondary Endpoints: |
Durable clinical benefit Time to progression Progression-free Survival Overall Survival Safety Patient-related outcomes |
| Target Sample Size: | 68 enrolled patients |
| Protocol Release Date: | 15 July 2022 |
Trial Organisation |
|
| Trial Chair: | Jarushka Naidoo, Dublin, Ireland |
| Trial Co-Chairs: |
Colin Lindsay, Manchester, United Kingdom Bartomeu Massuti, Alicante, Spain |
| Sponsor: | ETOP IBCSG Partners Foundation |
| Coordinating Group: |
ETOP IBCSG Partners Foundation |
| Participating Groups: |
Cancer Trials Ireland (CTI) and the Spanish Lung Cancer Group (SLCG) |
| Participating Countries: |
Belgium, France, Ireland, Italy, Spain, and United Kingdom |
| Registrations: |
EudraCT number: 2022-002736-31 clinicaltrials.gov: NCT05673187 |
Contact
Virginia Rodriguez Martinez (Clinical Trial Manager)
ETOP IBCSG Partners Foundation
Effingerstrasse 33
3008 Bern, Switzerland
- Details
|
|
A multicentre, randomised, phase II trial of brigatinib consolidation versus observation or durvalumab in patients with unresectable stage III NSCLC and ALK-rearrangement, after definitive chemo-radiotherapy.
BOUNCE: Brigatinib post definitive chemo-radiotherapy in patients with ALK-fusion non-small cell lung cancer
Trial Scheme
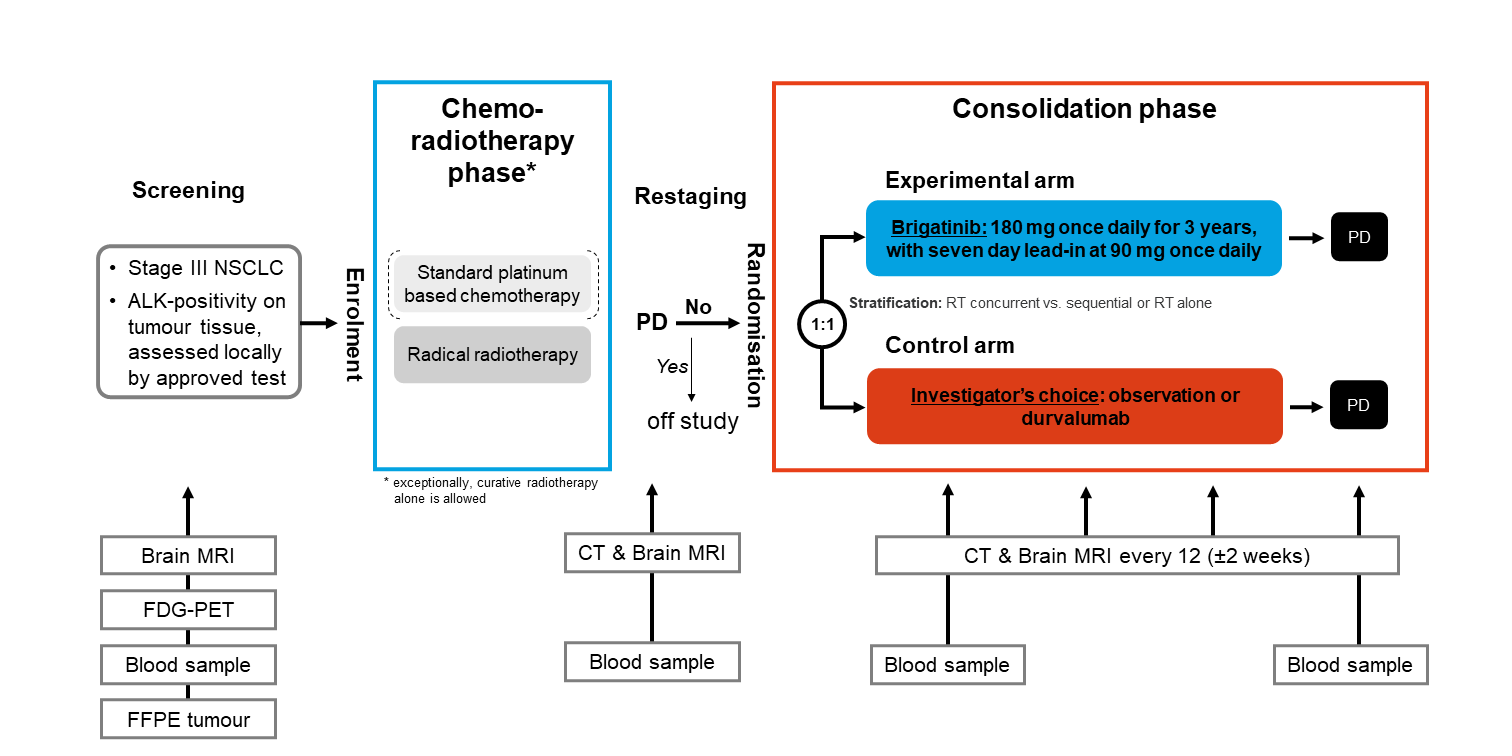
| Primary Endpoint: | Progression-free survival (PFS) according to RECIST v1.1 |
| Secondary Endpoints: |
Overall survival CNS-relapse-free survival Pattern of disease progression Toxicity according to CTCAE v5.0 |
| Target Sample Size: | 44 randomised patients (approximately 55 enrolled) |
| Protocol Release Date: | 6 February 2024 |
Trial Organisation |
|
|
Trial Chair: |
Rafal Dziadziuszko, Gdansk, Poland |
| Trial Co-Chair: |
Sanjay Popat, London, United Kingdom |
| Sponsor: | ETOP IBCSG Partners Foundation |
| Coordinating Group: | ETOP IBCSG Partners Foundation |
| Participating Groups: | SLCGT |
| Participating Countries: |
France, Italy, Poland, Spain, and the United Kingdom |
| Registrations: |
EU CT number: 2022-502467-38 clinicaltrials.gov: NCT05718297 |
Contact
Rico Hunkeler (Clinical Trial Manager)
ETOP IBCSG Partners Foundation
Effingerstrasse 33
3008 Bern, Switzerland


