|
|
Afatinib in pretreated patients with advanced NSCLC harbouring HER2 exon 20 mutations
NICHE: Afatinib in NSCLC with HER2 mutation
The erbB family of proteins comprises the receptor tyrosine kinase EGFR and the closely related HER2, HER3 and HER4. HER2 mutations are identified in about 2% of non-mall-cell lung cancers (NSCLC) and appear to be critical for lung carcinogenesis. Clinically relevant HER2 mutations are clustered in exon 20 of the HER2 gene and commonly include small exon 20 insertions, single amino acid or clustered substitutions.
Afatinib is a selective and irreversible small molecule inhibitor of the erbB family of proteins. It has been shown to effectively inhibit EGFR, HER2 and HER4 phosphorylation resulting in subsequent suppression of tumour growth.
The NICHE trial was designed to evaluate the ability of afatinib to control disease in pre-treated patients with advanced NSCLC harbouring HER2 exon 20 mutations.
Trial Scheme
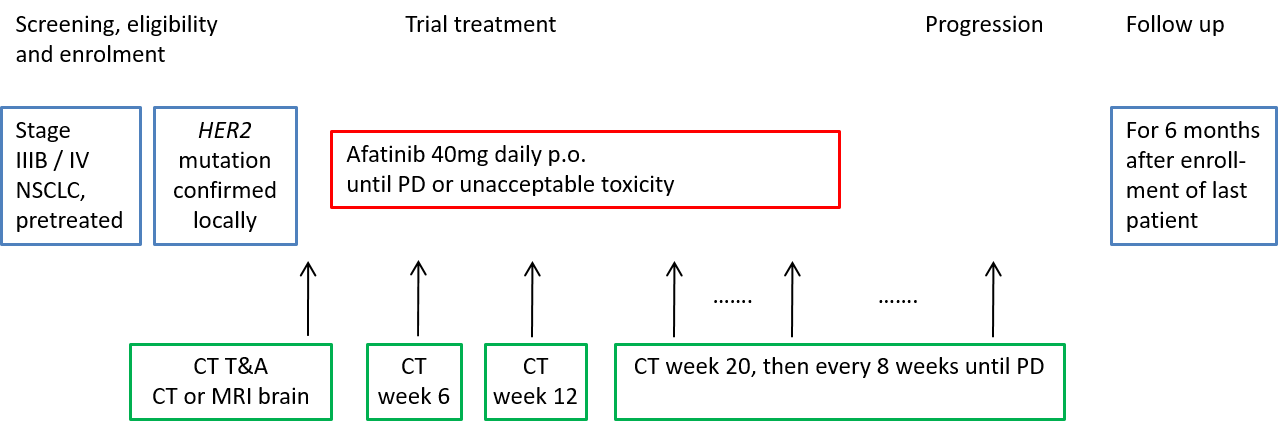
| Primary Endpoint: | Disease control lasting at least 12 weeks |
| Secondary Endpoints: |
Objective response Progression free survival Overall survival Toxicities of treatment |
| Target Sample Size: | 22 Patients (9 in stage I and 13 in stage II) |
| Final Accrual: | 13 Patients |
| Protocol Release Date: | June 2015 |
| Trial Activation Date: | 18 May 2015 |
| First Patient In: | 16 September 2015 |
| Accrual Closure Date: | 20 December 2016 |
| Global Trial Completion Date: | 17 August 2017 |
Trial Organisation |
|
| Trial Chairs: | Solange Peters and Rafal Dziadziuszko |
| Sponsor: | ETOP |
| Coordinating Group: | ETOP |
| Participating Countries: |
Germany, Netherlands, Spain, and Switzerland |
| Registrations: |
EudraCT number: 2014-005098-35 ClinicalTrials.gov: NCT02369484 |
Contact
ETOP IBCSG Partners Foundation
Effingerstrasse 33
3008 Bern, Switzerland


